Canvas Dx: AI Powered App To Diagnose Autism Gets FDA Marketing Authorization
Back in October 2020, Cognoa, a company that makes digital therapeutics and diagnostic tools, submitted an Artificial Intelligence powered tool for FDA approval that aims to help diagnose Autism in children. Just last week, Cognoa received approval from FDA for it’s Canvas Dx software and will begin to market this software later this year.
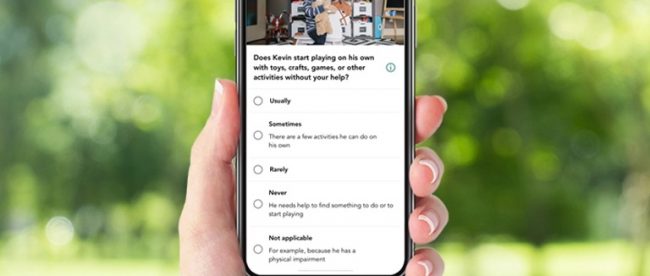
The app asks parents to answer a questionnaire related to their child’s behavior patterns and also upload videos of their child in their natural environment. Once submitted, the responses and videos are sent to a trained professional who observes the responses and videos and provides their own input. The child’s pediatrician also provides their responses about the child’s behavior.
On average, most kids are diagnosed around the age of four but the goal is to diagnose them much sooner – at the age of eighteen months, so they can have access to services like speech therapy as soon as possible. The current system is unable to provide early diagnoses to many children and families.
The FDA approval comes after an evaluation done on 425 kids aged 18 months to five years across diverse a group of patients in terms of race, gender, education, and socioeconomic status.
Head to the post from last year to learn more about this app. You can also sign up to get updates on Canvas Dx.
Source: PR Newswire, MedCityNews

Thank you for sharing this wonderful post